 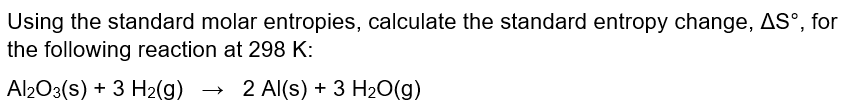
After the reaction, the two are bonded together and can't float around freely from one another. In other words the N 2( g) used to float around independently of the H 2 gas molecules. This is expected because we are decreasing the number of gas molecules. It would appear that the process results in a decrease in entropy - i.e. \įrom the balanced equation we can write the equation for ΔS 0 (the change in the standard molar entropy for the reaction): As with other calculations related to balanced equations, the coefficients of each component must be taken into account in the entropy calculation (the n, and m, terms below are there to indicate that the coefficients must be accounted for): The entropy change in a chemical reaction is given by the sum of the entropies of the products minus the sum of the entropies of the reactants. Unlike enthalpies of formation, standard molar entropies of elements are not 0.When comparing standard molar entropies for a substance that is either a solid, liquid or gas at 298 K and 1 atm pressure, the gas will have more entropy than the liquid, and the liquid will have more entropy than the solid.A table of standard molar entropies at 0K would be pretty useless because it would be 0 for every substance (duh!) Standard molar entropy values are listed for a variety of substances in Table T2. the entropy of a pure substance at 298 K and 1 atm pressure). Standard molar entropies are listed for a reference temperature (like 298 K) and 1 atm pressure (i.e.The entropy of a substance has an absolute value of 0 entropy at 0 K. E) It is endothermic., How does the third law of thermodynamics allow absolute entropies of substances to be determined A) It defines a reference point by. In fact, values for the "standard molar entropy" of a substance have units of J/mol K, the same units as for molar heat capacity. Study with Quizlet and memorize flashcards containing terms like Which of the following is true for a process where S(universe) < 0 at 298 K A) It is exothermic B) It is nonspontaneous. the rise in temperature is the heat capacity, it would seem that in some way, information about the heat capacity (and how it changes with temperature) would allow us to determine the entropy change in a system. Since the quantitative term that relates the amount of heat energy input vs. The absolute entropies for X,Y and Z are 120 J K1mol1, 213.8 J K1 mol1 and 197.9 J K1mol1 respectively. all the ice has melted or all the liquid has frozen) However, in both of the above situations, the energy change is not accompanied by a change in temperature (the temperature will not change until we no longer have an equilibrium condition i.e. system J/K ASO Consider the reaction: 2N2(g) + O2(g) 2N2O(g) Using standard absolute entropies at 298K, calculate the entropy change for the system when 1. Likewise if a small amount of energy is withdrawn from the system, the equilibrium will shift to the left (more ice) Question: Consider the reaction: CO2(g) + H2(g) CO(g) + H2O(g) Using standard absolute entropies at 298K, calculate the entropy change for the system when 2.38 moles of CO2(g) react at standard conditions.If a small amount of energy is input into the system the equilibrium will shift slightly to the right (i.e.At such a temperature and pressure we have a situation (by definition) where we have some ice and some liquid water.\) illustrates this procedure for the combustion of the liquid hydrocarbon isooctane (C 8H 18 2,2,4-trimethylpentane).\] 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
